
We get our answer when we see a neutral, neutral state or an electric point. It will be 6 plus 9.17 divided by 2, and that will give us a partial calculator. Complete step-by-step answer: If an amino acid has only one amino acid and. The pi is going to be the 2 peaks on the side of the neutral atom divided by 2 points. (2) At the isoelectric point, the zwitterions form of the amino acid is dominant. I try to circle the two positive charges that were there to make it more obvious. At pH values above or below the isoelectric point, the molecule will have a net charge which depends on its pI value as well as the pH of the solution in which the amino acid is found. Each amino acid has its own pI value based on the properties of the amino acid. 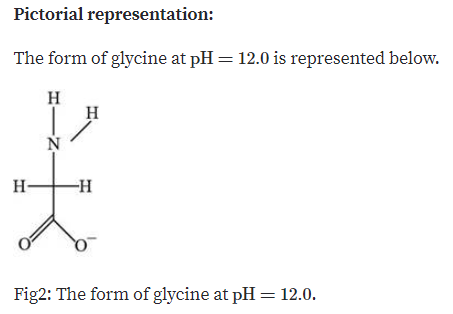
Group is what it is, and we are going to protenate the car box over right. The zwitterion of an amino acid exists at a pH equal to the isoelectric point. When it's petatone the periodic table, not a periodic, an amen acid table, and we have this positive charge on that mines, I always see a lot of times. If we go to the right, we'll lose the positive charge and still have a negative charge on our next one. We're going to be neutral 1 because they out. For a histedine, I'll be like this and histedene is a complicated ring structure, and we see it as a neutral charge in the muni group. The various phes are ph's of 148 and 11 point, so we're going to have 4 structures total, and I'm going to start at our neutral state because we're going to have a positive charge here and a neutral. At intermediate pHs the zwitterion concentration increases, and at a characteristic pH, called the isoelectric point (pI), the negatively and positively. Since different sources provide different values of pK values, we are averaging the isoelectric point values that are obtained using pKs from the following sources: Wikipedia. The calculation is based on the pK values of the amino acids and of the terminal amine & carboxyl group of the peptide chain. At this pH the the amino acid sequence forms a zwitterion. It is represented by the formula p I p K a 1 + p K a 2 2. The isoelectric point is defined as the pH at which a particular amino acid sequence (peptide chain) bears no net electrical charge. Select an amino acid, then drag the pH arrow around to see. The pH value at the isoelectric point can be calculated from the equilibrium constants (acid and base) of the Zwitterion. Just like 9.17, we have it at 9.17, and that will give us our various peka values of peka, 1 or just pek 1 or right. It will be close to 2, but not quite our next one, it is a nice whole number. We have our p ka values at 1.829.7 and 6 for our group and these will take place that are half equivalents like 1.5, 2.5, and 1 so for our lowest, 1 will put it at.5. We're going to draw a curve for hestidene that's 2.511.528.53 and we're going to draw our structures of various phis on top of it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
